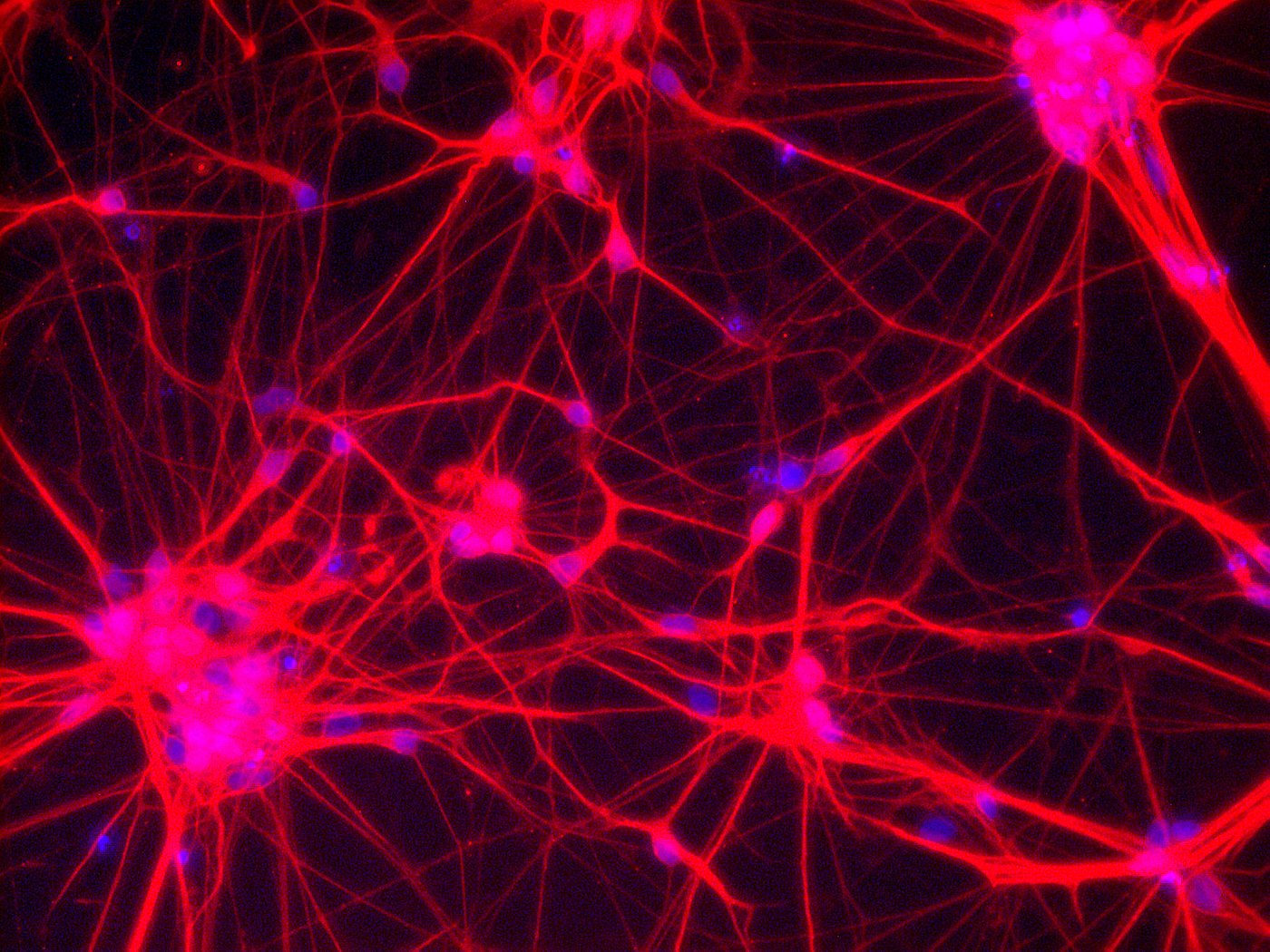
Neurons – Network of neuronal cells (Blue: nucleus; Red: neurons) [Copyright: Institute for Transfusion Medicine, UK Essen]
Stem cell research is associated with great hope for new therapeutic approaches - but so far, only a few stem cell-based treatments have been approved. The research and approval of therapies is a complex process that requires strict rules for the benefit of patients. But do the existing laws fit the state of the art in research, and are they compatible with the practice of the "bench to bedside" process, i.e. the transfer of research results from the laboratory to the treatment of patients? To address these questions, the Stem Cell Network NRW commissioned two legal opinions from Prof. Dr. iur. Dr. rer. pol. Tade M. Spranger (University of Bonn) and Prof. Dr. iur. Ulrich M. Gassner (University of Augsburg), which were completed in 2019 and form the basis of the new book.
In the first part, Professor Spranger analyzes, among other things, the regulation of research on human induced pluripotent stem cells as well as the translation prohibition of the Stem Cell Act. He also sheds light on the interaction of new genome editing technologies (CRISPR/Cas9) with existing regulations.
In the second part of the volume, Professor Gassner examines the need for regulation in the transfer of research results into therapeutic and diagnostic applications. In particular, the requirements for the approval of stem cell therapeutics and the granting of manufacturing authorizations are discussed.
The volume has been published with Nomos in the series "Schriften zum Bio-, Gesundheits- und Medizinrecht" (in German) and can already be ordered.
Bibliographic information:
Gassner, Ulrich M. & Tade M. Spranger (2020): Stammzellen in Forschung und Therapie: Rechtsrahmen und Reformbedarf; Schriften zum Bio-, Gesundheits- und Medizinrecht; Band 43; Nomos.
ISBN: 9783848768646